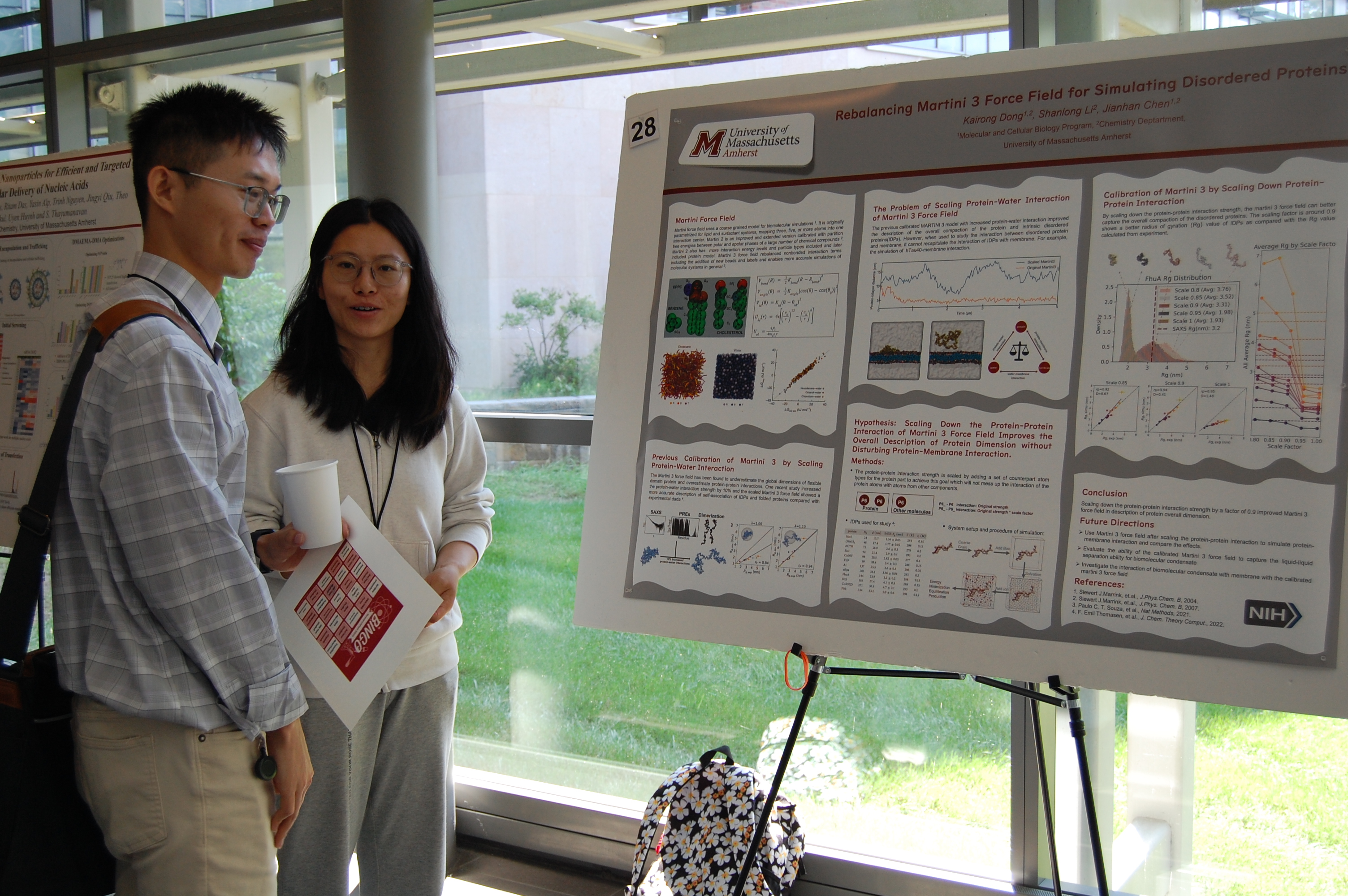
The annual ResearchFest in the Chemistry department is almost the biggest event of the whole year. In the ResearchFest event, graduate students from the department will participate in poster presentations or luckily get selected as the key presentators to present our fascinating research. Judges, usually our dear faculty members, will grade the quality of the presentation and science for all presentations, based on which students will win awards. Super fun! You can get more information from: UMass Chemistry ResearchFest.
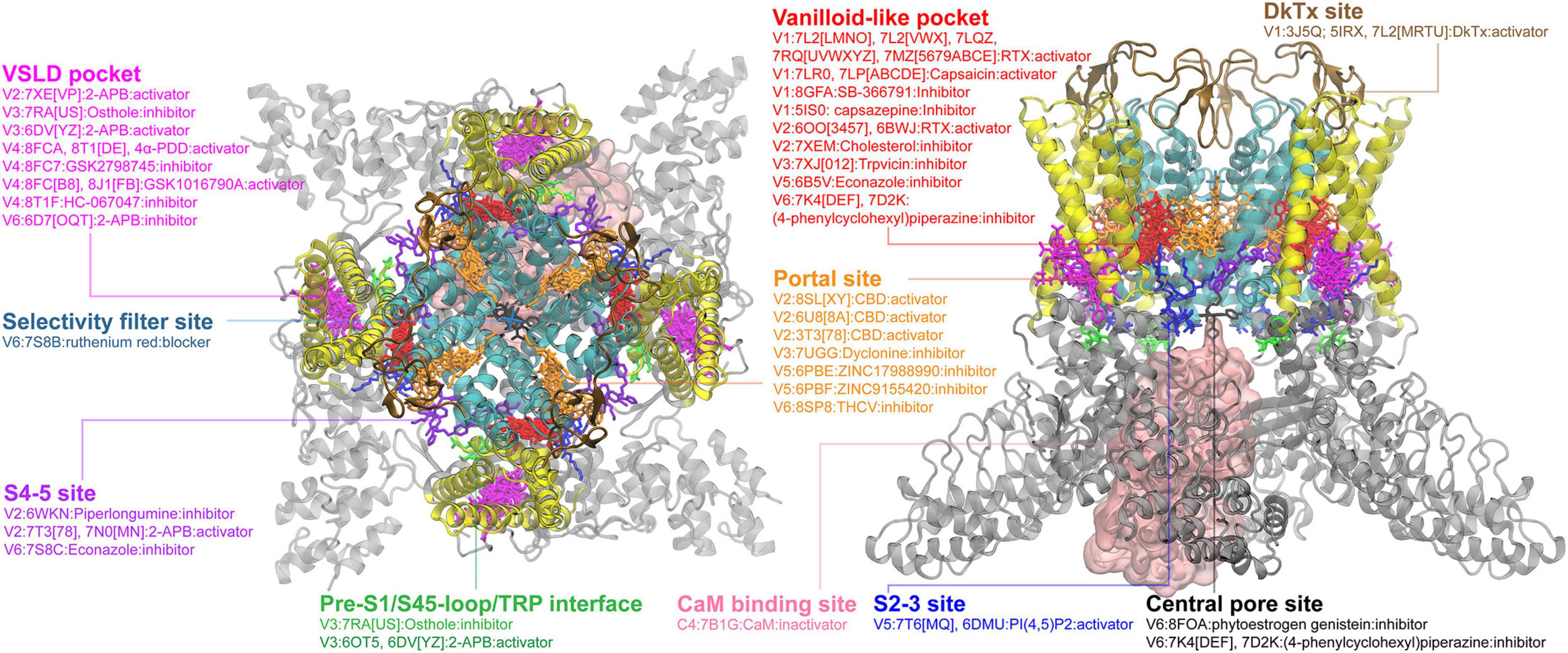
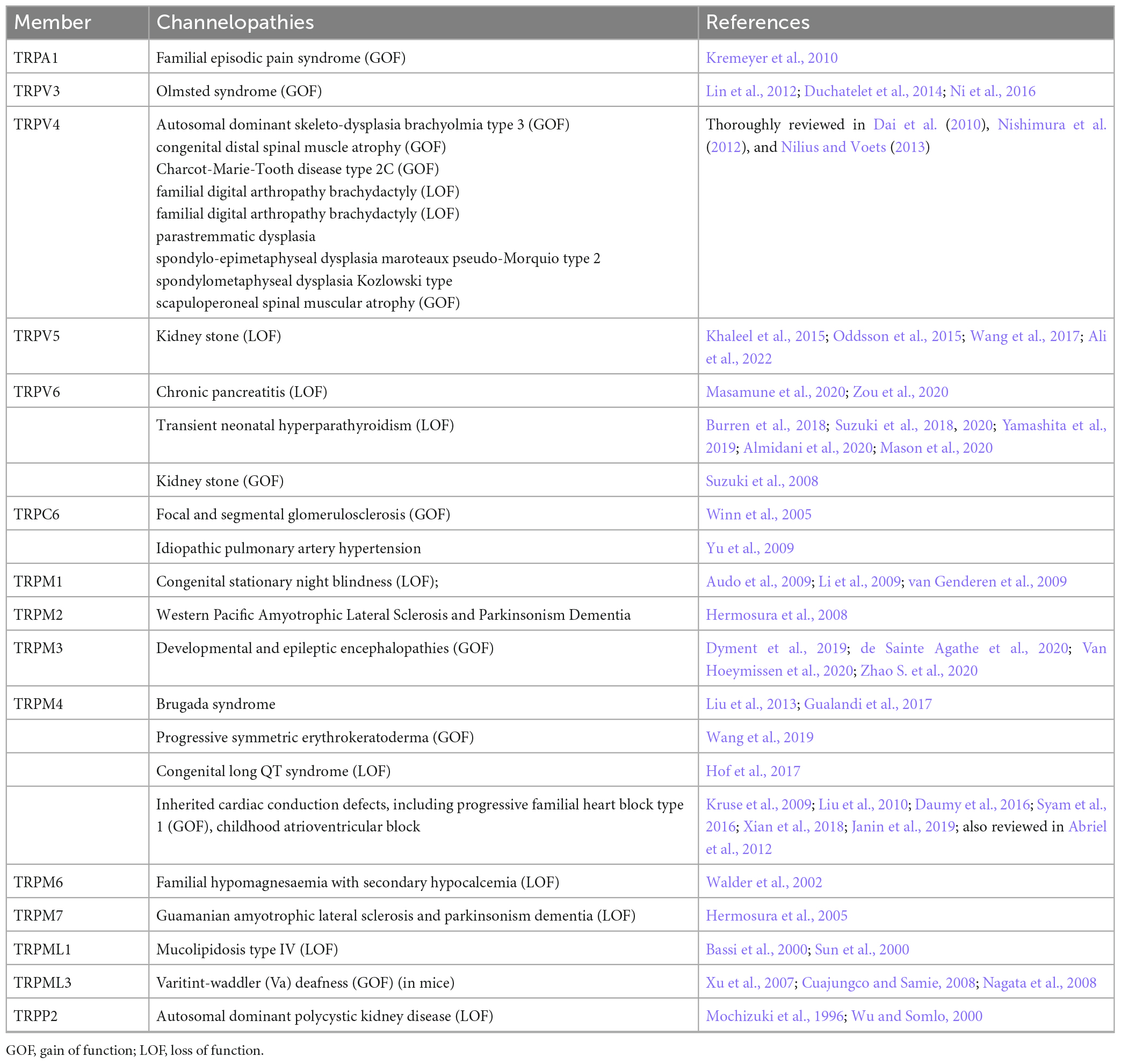
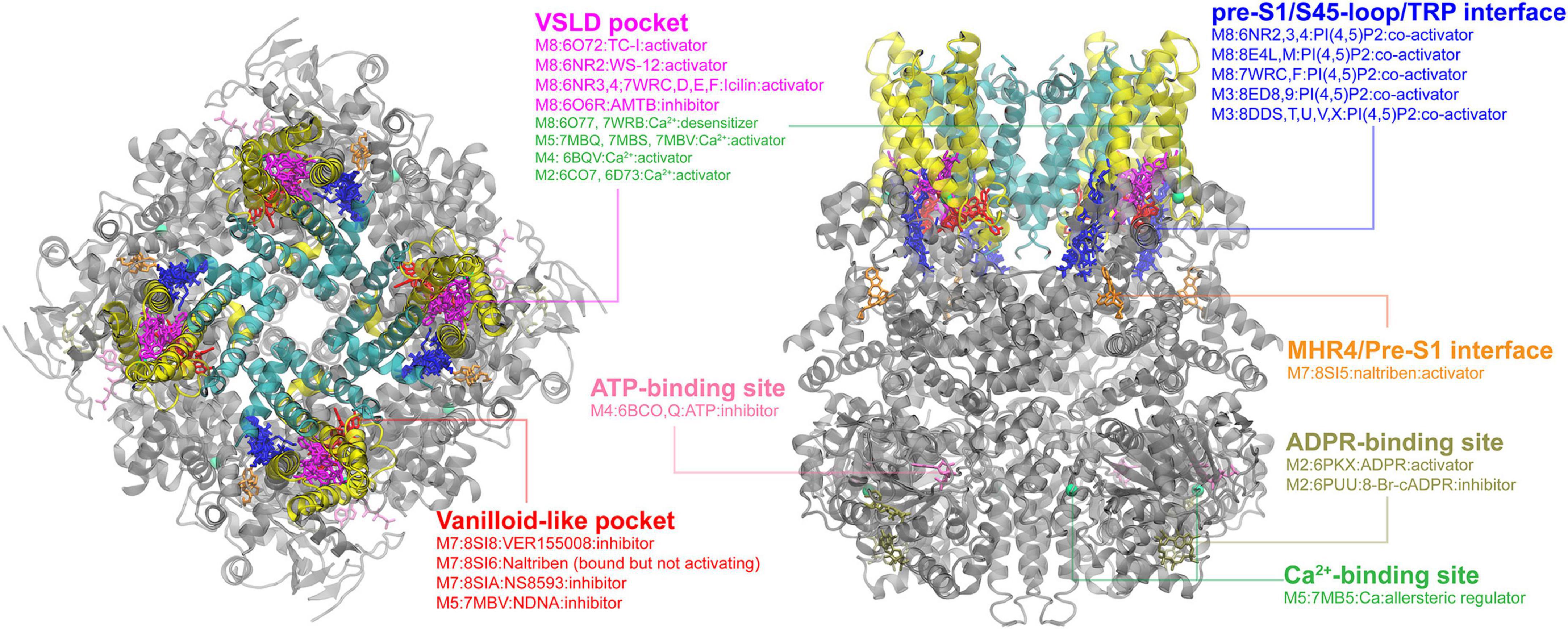
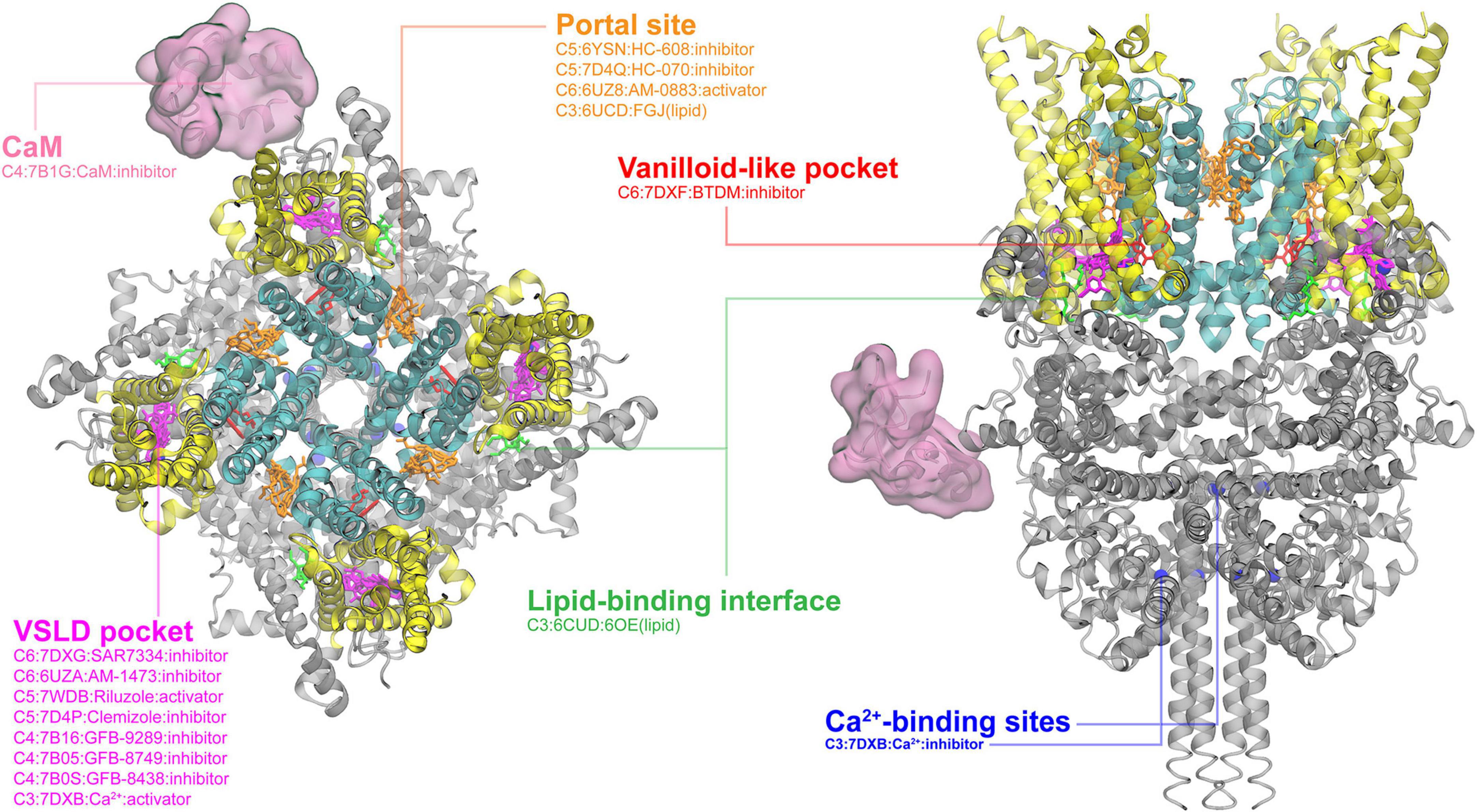
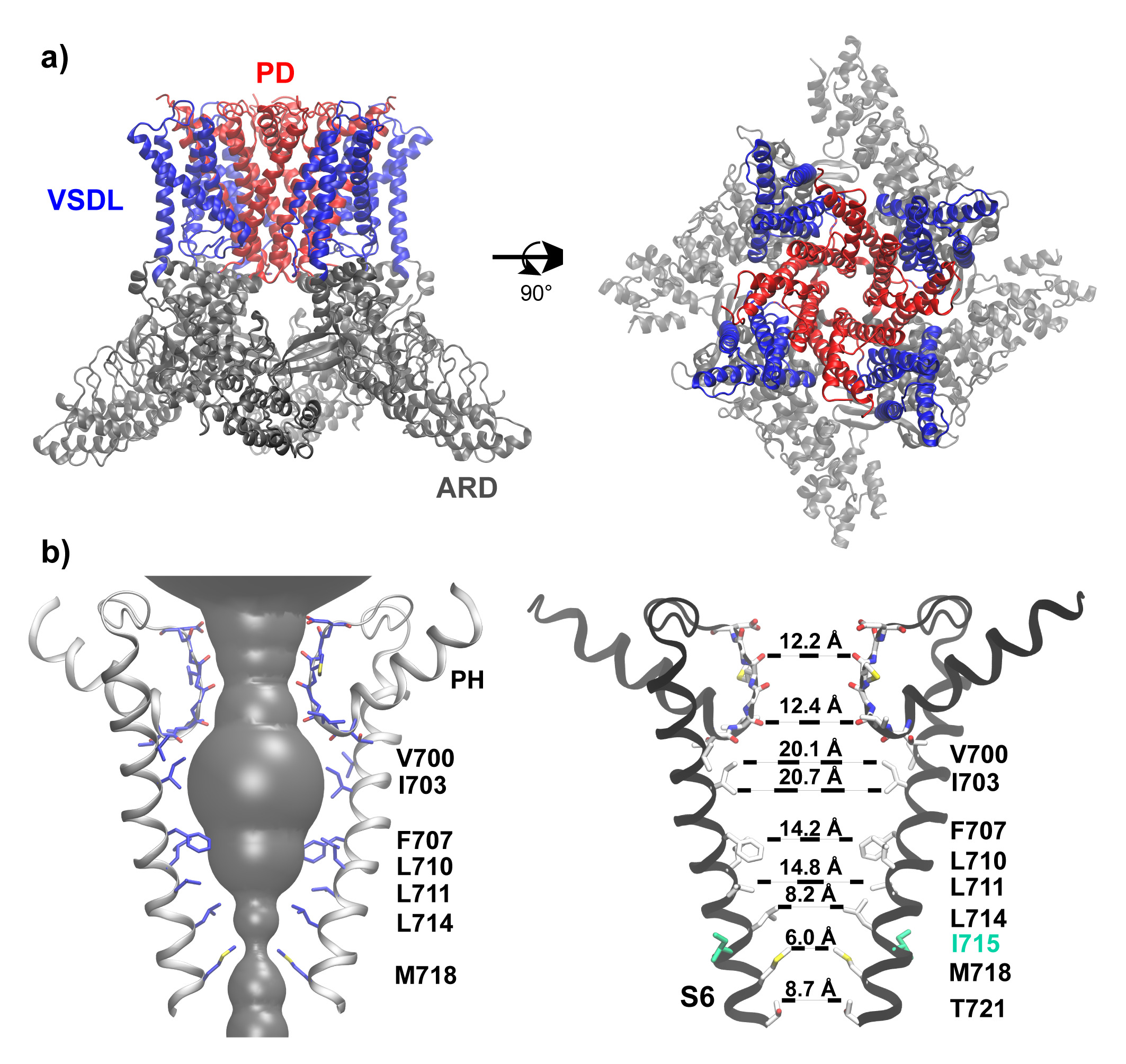
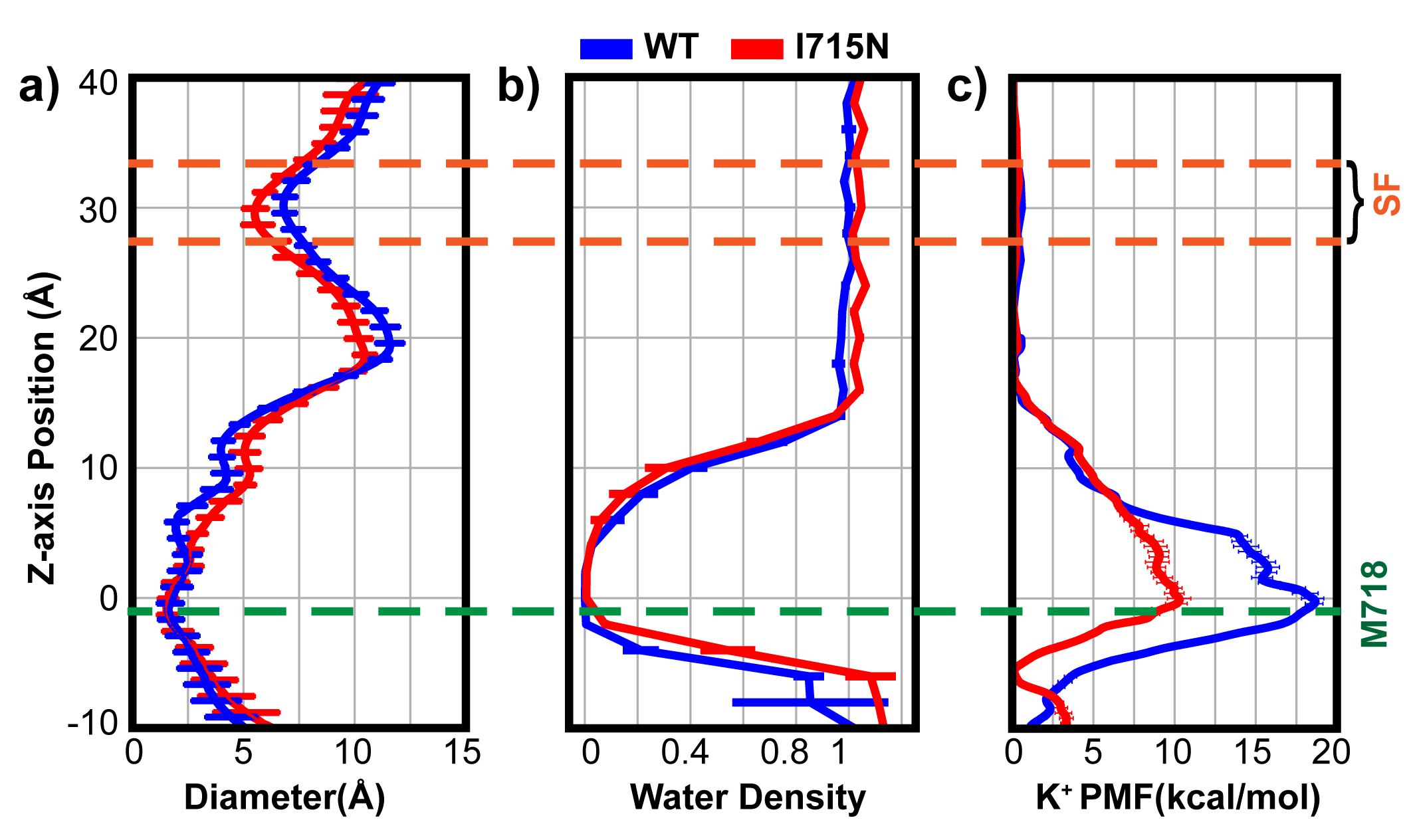
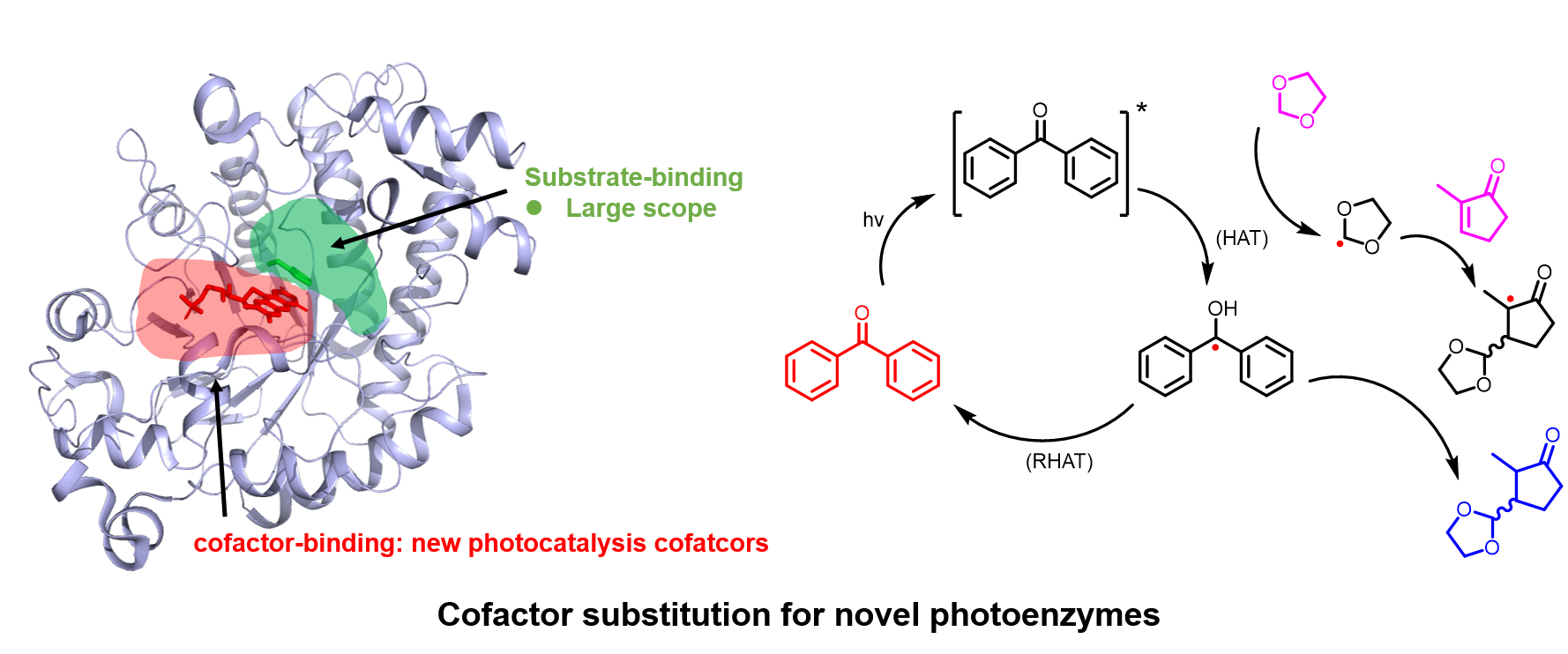
In the event this year, I was selected as one of the key presentator to give a presentation to the whole department, including new coming graduate students. I focused the talk on the gating mechanisms of ion channels.
Jian in the presentation
Jian was awarded the 2nd place for the presentation
I also participated in the post presentation section as one of the many audience. Always nice to hear people talking about exciting science projects.
The 2021-batch graduate students
I really want to thank my professor Jianhan Chen who always gives me great suggestions and provides me with many opportunities; my mentor Mahdieh Yazdani (during merck internship) who help me with my researchfest presentation, and all my labmates who have always been supportive.
]]>